Carbon dioxide-mediated thermochemical conversion of banner waste using cobalt oxide catalyst as a strategy for plastic waste treatment
Nahyeon Lee, Kun-Yi Andrew Lin and Jechan Lee
Abstract
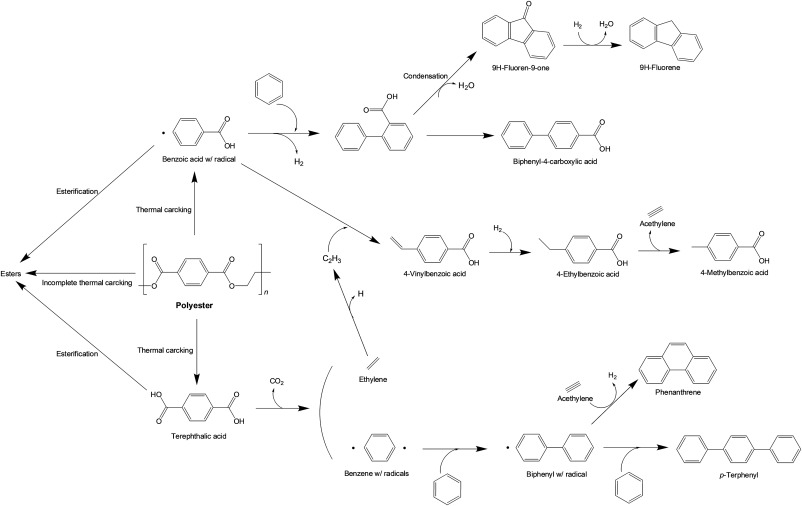
In this study, the effects of CO2 thermochemical agent and a metal oxide catalyst (Co3O4) on thermochemical banner waste conversion were explored. The results revealed that compared to the non-catalytic conversion of banner waste under N2 environment, the conversion under CO2 yielded more non-condensable gases owing to an enhanced thermal cracking of volatiles. In addition, the CO and CH4 yields at >700 °C in CO2 increased considerably owing to the reverse water–gas shift reaction and CO2 methanation. The CO2 agent reduced the yields of condensables (e.g., benzoic acids, phthalic acids, esters, biphenyls, fluorenes) and decomposition residue (e.g., char and wax), which could be attributed to the enhancement of the thermal cracking of volatiles evolved during the banner waste conversion by CO2 and the C–H and O–H bonds present in the feedstock. In addition, the Co3O4 catalyst promoted the decarboxylation reaction under N2 environment, whereas it promoted the methanation and reverse water–gas shift reaction under CO2. This indicates that compared to the non-catalytic CO2-assisted banner waste conversion, the use of CO2 for the conversion of banner waste in the presence of Co3O4 significantly increased the yields of CH4 and CO. Furthermore, Co3O4 promoted the thermal cracking of polyester bond, thus decreasing the yields of long-chain chemical compounds. In addition, the simultaneous use of Co3O4 catalyst and CO2 agent minimized the formation of char and wax. For all cases (N2 versus CO2, non-catalytic versus catalytic), an increase in temperature enhanced the total permanent gas yield and decreased the yields of condensables, char, and wax. The findings of this study revealed the importance of the synergistic use of Co3O4 catalyst and CO2 agent for the plastic waste upcycling, such as banner waste.
